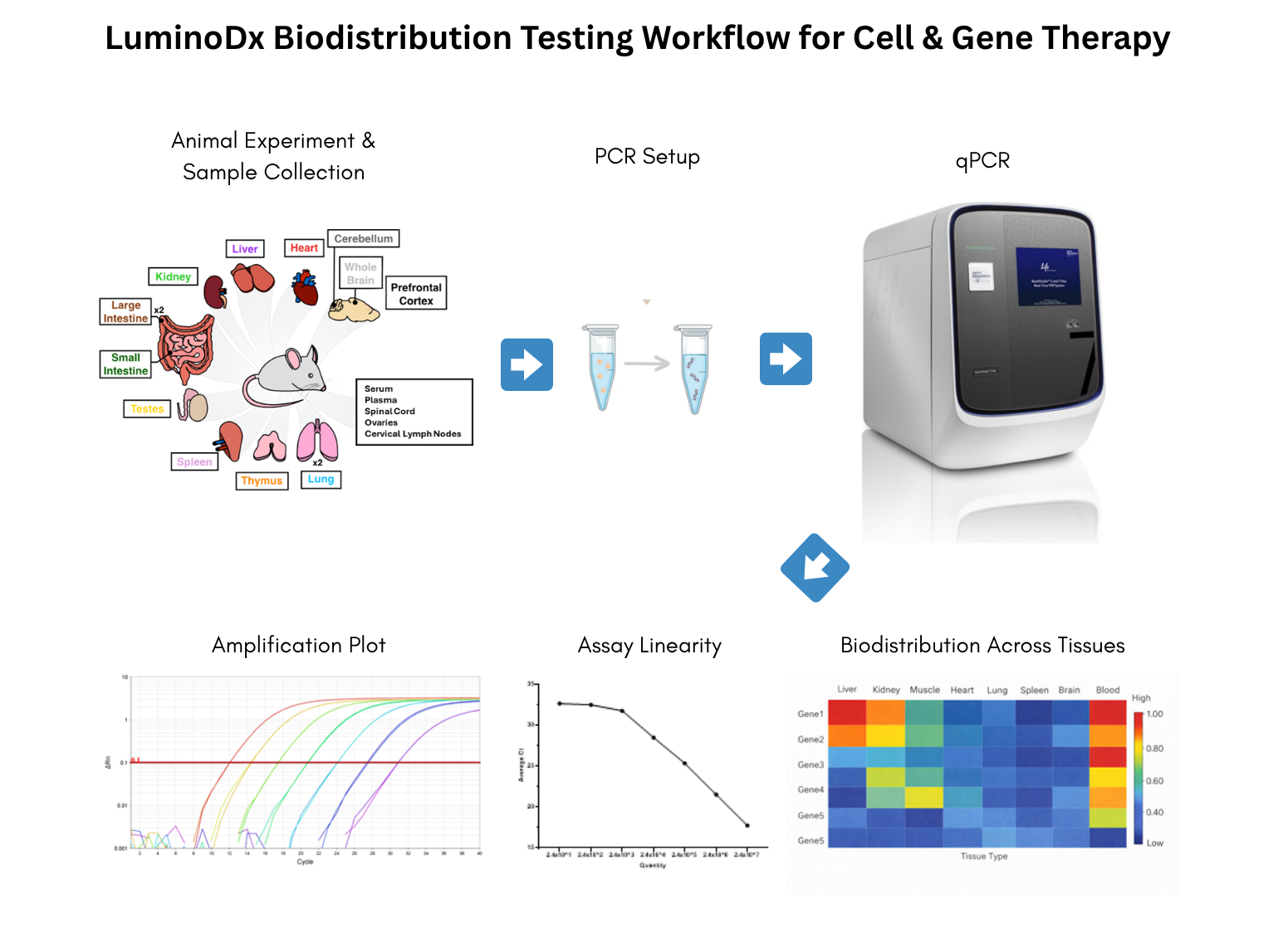
Nucleic acid extraction will be carried out with analytically validated methods from different tissue types (e.g., liver, spleen, brain) to ensure high yield, purity, and minimal PCR inhibitors.
Assay Design: Primers and Probes will be selected specific to the test article (e.g., the target’s DNA sequence) to prevent cross-reactivity with host DNA or other related sequences.
Standard Curve: A calibration curve with known concentrations of the target nucleic acid will be formulated (e.g., a plasmid) in a background of host DNA. This curve will be used to quantify the test article in unknown study samples.
Molecular assays used in Good Laboratory Practice (GLP) biodistribution studies undergo rigorous analytical validation.
The following key parameters will be assessed during the formal analytical validation:
All steps of the validation process will be thoroughly documented to meet GLP principles. This includes:
Once validated, the molecular assay will be considered “fit for purpose” and can be used to analyze study samples from the biodistribution study, with ongoing quality control checks included in each run.
For Quality, expedient results for your submissions please consider LuminoDx!